Choosing a red light therapy panel for your brand involves more than comparing LED counts and price. The six criteria that determine long-term product success are:
- Configuração de comprimento de onda — does it go beyond basic 660nm + 850nm?
- Irradiance data transparency — does the supplier provide multi-distance measurements?
- Control system and user experience — touchscreen, APLICATIVO, escurecimento, and mode flexibility
- Customization depth — logo-only private label vs. full OEM/ODM development
- Certification coverage by market — matched to your target country and channel
- Factory capability and process structure — EVT/DVT/PVT, cross-functional review, change management
Each criterion is covered in detail below, with specific questions to ask suppliers and red flags to watch for.
Introdução: Why Most Panel Comparisons Start in the Wrong Place
Walk into any sourcing conversation for red light therapy panels, and the first question is almost always the same:
“How many LEDs does it have?”
LED count is visible. It is easy to compare. It fits neatly into a specification table.
The problem is that LED count does not predict whether your product will succeed in the market. It does not tell you whether the wavelength design will support your brand positioning. It does not tell you whether the irradiance data will hold up when a distributor asks for proof. It does not tell you whether the supplier can actually customize the product to match your brand identity, your target market’s compliance requirements, or your customers’ expectations for a smart, premium device.
Most red light therapy panels on the market today look very similar on a spec sheet. Many offer the same two or three wavelengths, similar LED counts, similar power ratings, and similar price ranges. For brands trying to build a differentiated product line, this creates a real challenge.
The question is not which panel has the most LEDs. The question is which panel — and which manufacturer — gives your brand the strongest foundation for long-term product success.
Na Wakelife Beauty, depois 10+ years of manufacturing medical-grade LED light therapy devices for 1,000+ global brand partners across the US, UE, Reino Unido, Austrália, e Japão, we have seen a consistent pattern: the brands that succeed make sourcing decisions based on criteria that go beyond the surface specification. The brands that struggle usually focus on price and LED count — and discover the other factors later, at a much higher cost.
This article covers the six criteria we recommend evaluating before choosing a red light therapy panel for your brand, with specific questions to ask suppliers at each stage.
Criterion 1 — Wavelength Configuration
Why This Matters More Than LED Count
Wavelength is the core technical parameter of any red light therapy product. It determines the type of light the device emits, how the light interacts with tissue at different depths, and — critically for brands — what product story you can credibly tell.
Most entry-level and mid-range panels are built around two wavelengths: 660nm red light and 850nm near-infrared light. This combination is widely used, well-documented, and commercially proven. It is also the configuration that almost every competitor offers.
For brands launching in 2025 and beyond, a 660nm + 850nm panel is a starting point — not a differentiator.
The Three Tiers of Wavelength Configuration
Basic configuration: 660nm + 850nm The market standard. Easy to source, easy to explain, easy to compare — and therefore difficult to differentiate. If your primary strategy is price competition or marketplace volume, this configuration is sufficient. If your strategy involves premium positioning, credibilidade clínica, or product line expansion, you will likely hit its limitations quickly.
Intermediate configuration: 630nm + 660nm + 810nm + 830nm + 850nm Adding shorter red wavelengths (630nm) and additional NIR wavelengths (810nm, 830nm) gives the product a broader light spectrum and more flexibility for different use scenarios. This configuration supports a stronger technical story without requiring a major cost premium, and gives brands more dimensions to explain in product pages, user education content, and distributor materials.
Advanced configuration: 630nm + 660nm + 810nm + 830nm + 850nm + 1060nm The addition of 1060nm deep near-infrared is the most significant differentiator available in current panel products. 1060nm is positioned at the deeper end of the NIR spectrum, and its inclusion allows brands to communicate a meaningfully different product architecture — not just a variation on the same basic formula.
For brands targeting premium wellness, profissional, or clinical-adjacent markets, 1060nm is one of the few wavelength additions that creates a genuine technical separation from standard panels.
Questions to Ask Your Supplier
- What is the exact wavelength for each LED type, and what is the binning tolerance (Por exemplo, ±3nm vs. ±10–15nm)?
- Can the wavelength configuration be customized for OEM/ODM projects?
- Is 1060nm available as a standard configuration, or does it require custom LED sourcing?
- How are wavelength combinations organized across dimming channels?
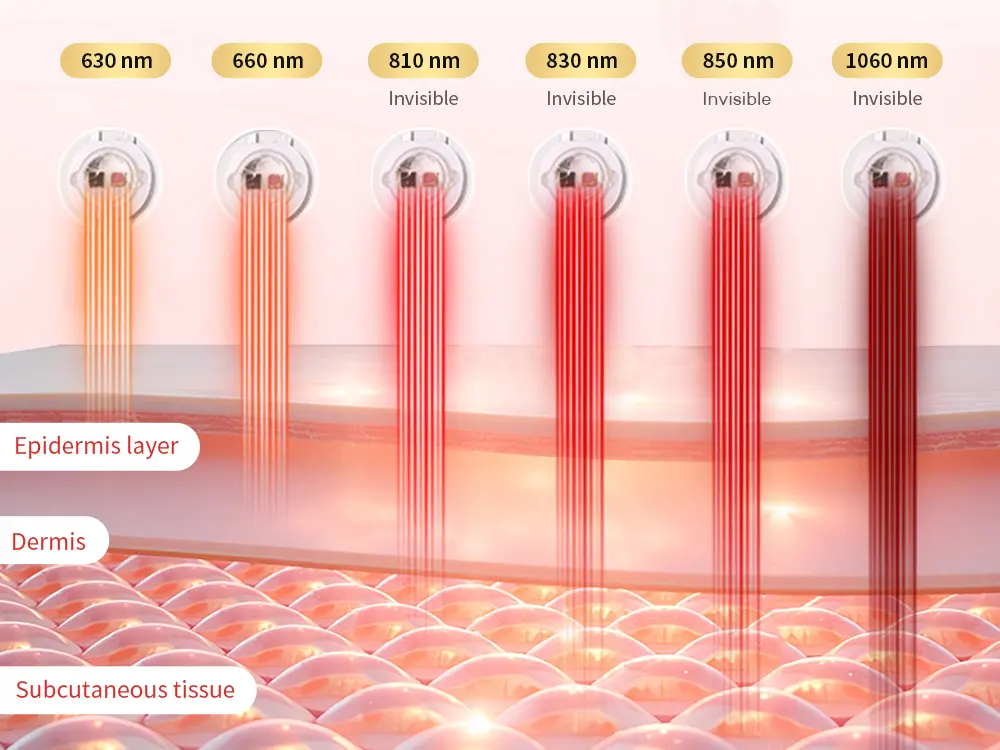
Wakelife Beauty’s ESPLUS750 panel uses a six-wavelength configuration: 630nm / 660nm / 810nm / 830nm / 850nm / 1060nm, entregue através 150 LEDs de chip duplo. This configuration gives OEM/ODM brand partners a technical foundation that supports premium product positioning across beauty, bem-estar, recuperação, and professional equipment markets — and a product story that goes significantly further than standard two-wavelength panels.
Criterion 2 — Irradiance Data and Transparency
Why LED Count Is Not a Performance Metric
LED quantity appears on almost every red light therapy panel specification sheet. It is also one of the least meaningful numbers for predicting real-world light output performance.
Irradiance — the actual light energy delivered to the treatment area, medido em miliwatts por centímetro quadrado (MW/cm²) — is what matters. And irradiance depends not only on LED count, but on LED chip quality, optical lens angle, driver circuit stability, gerenciamento térmico, and measurement methodology.
A panel with 300 LEDs and poor thermal management may deliver lower sustained irradiance than a well-engineered panel with 150 LEDs and a stable driver circuit.
Para marcas, this matters in two ways. Primeiro, it affects the actual performance of the product your customers receive. Second, it affects your ability to make credible, defensible claims on your product page, in distributor materials, and in any regulatory documentation you may need for your target market.
What Irradiance Data Should Look Like
A transparent supplier should provide irradiance measurements at multiple distances — not just a single “peak” figure measured at the closest possible point.
For a mid-size panel like ESPLUS750, useful irradiance data includes:
- Measurement at 3 polegadas (close-range, higher intensity use)
- Measurement at 9 polegadas (standard home use distance)
- Measurement at 12 polegadas (wider coverage, lower intensity)
This gives brands accurate information for product positioning, and gives buyers a realistic picture of what the device delivers at the distances their customers will actually use.
Red Flags to Watch For
- Irradiance figures provided without specifying measurement distance
- Single-point measurements only (usually the closest possible distance)
- No test equipment or methodology disclosed
- Figures that cannot be independently verified
Questions to Ask Your Supplier
- Can you provide irradiance measurements at 3 polegadas, 9 polegadas, e 12 polegadas?
- What test equipment and methodology is used for irradiance measurement?
- Is irradiance consistent across the panel surface, or does it vary significantly between center and edges?
- How does irradiance hold up over extended use sessions (thermal stability)?
The ESPLUS750 Example
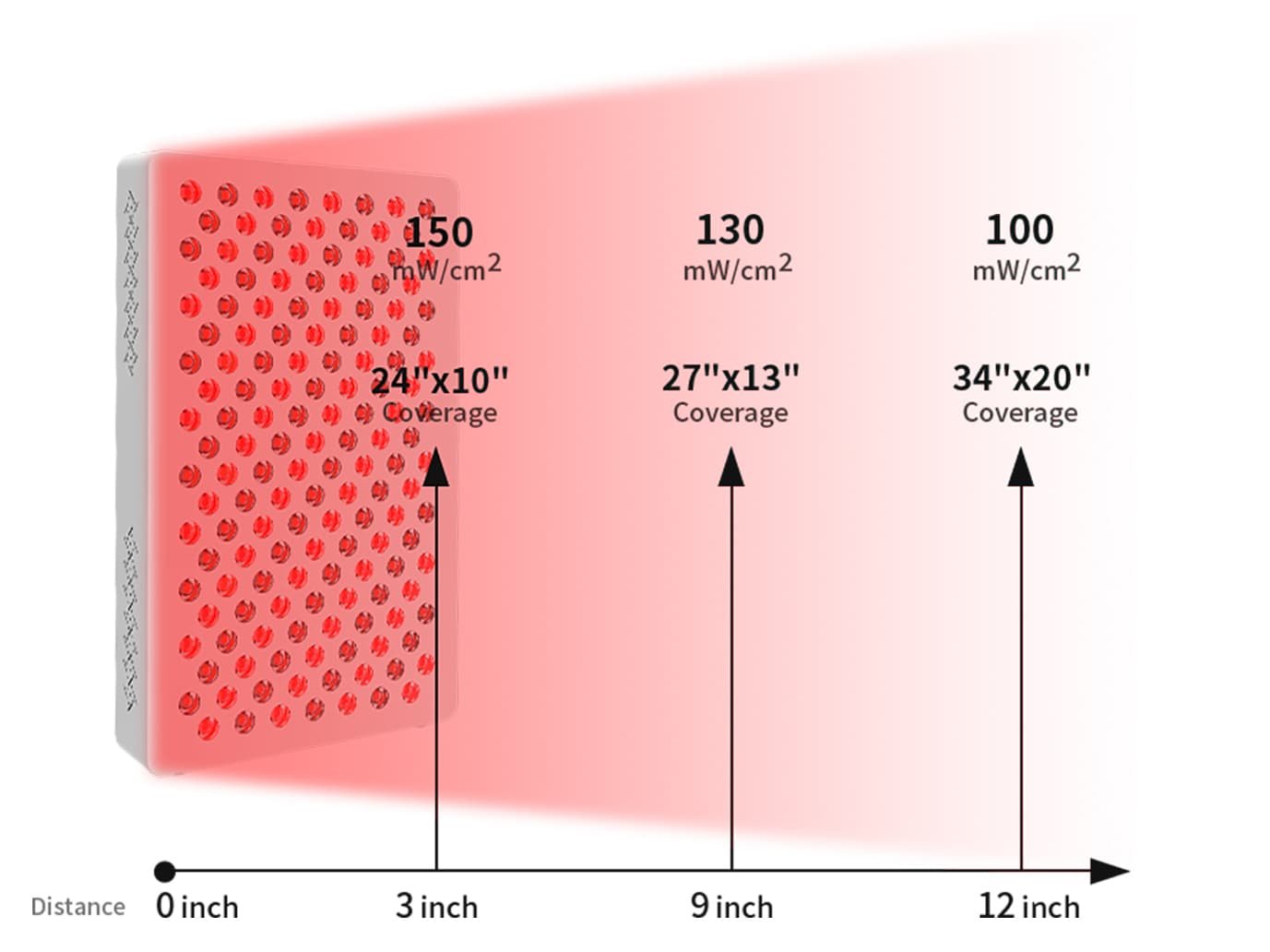
ESPLUS750 provides measured irradiance of 150em mW/cm² 3 polegadas, 130em mW/cm² 9 polegadas, e 100em mW/cm² 12 polegadas. These multi-distance figures give brand partners clear, usable performance data for product pages, buyer comparison materials, and internal sourcing evaluation — without requiring additional third-party testing before a sourcing decision can be made.
Criterion 3 — Control System and User Experience
Why Smart Control Is Now a Baseline Expectation
Consumer expectations for wellness devices have shifted significantly. A product that turns on and off — and does nothing else — is increasingly difficult to position at a premium price point, even with strong wavelength specifications and irradiance data.
Modern users expect:
- Intuitive, easy operation without reading a manual
- Personalized settings that can be saved and recalled
- Flexible timing and brightness control
- An experience that feels premium relative to the price
For brands targeting home wellness, cuidados de beleza, professional studio, or any market segment above the entry level, the control system is a meaningful part of the product value proposition — not a secondary detail.
The Control System Spectrum
Básico: Remote control only, or simple button panel. Suitable for entry-level price points. Limited user engagement and no smart device integration.
Intermediate: Touchscreen display with direct on-device control. Improves usability and perceived product quality without requiring app connectivity.
Avançado: Controle de aplicativos + tela sensível ao toque, with dimming, seleção de modo, custom settings, and multi-device management. This configuration creates a significantly more premium user experience and opens the door to brand-specific digital touchpoints — notifications, rastreamento de uso, program guidance — depending on the app customization level.
Dimming and Mode Architecture
Two features that are often underestimated in sourcing decisions:
Multi-channel dimming allows users to adjust individual wavelength groups independently, rather than controlling all LEDs at a single brightness level. For brands with a multi-wavelength panel, this is the feature that makes the wavelength configuration actionable — users can actually control the light mix, not just turn it on.
Modos predefinidos + modos personalizados serve two different user needs. Preset modes give new users a simple starting point. Custom modes give engaged users a reason to continue using the product on their own terms. For brand retention and repeat purchase, the combination of both is significantly more powerful than either alone.
Questions to Ask Your Supplier
- Does the panel support APP control? Is the APP already developed, or would it need to be built?
- Can the APP interface, mode names, and brand color system be customized for OEM projects?
- How many dimming channels are supported, and how are they organized across wavelength groups?
- How many preset and custom modes are available? Can mode names be adapted for different markets?
- Does the panel support multi-device connection for scalable setups?
The ESPLUS750 Example
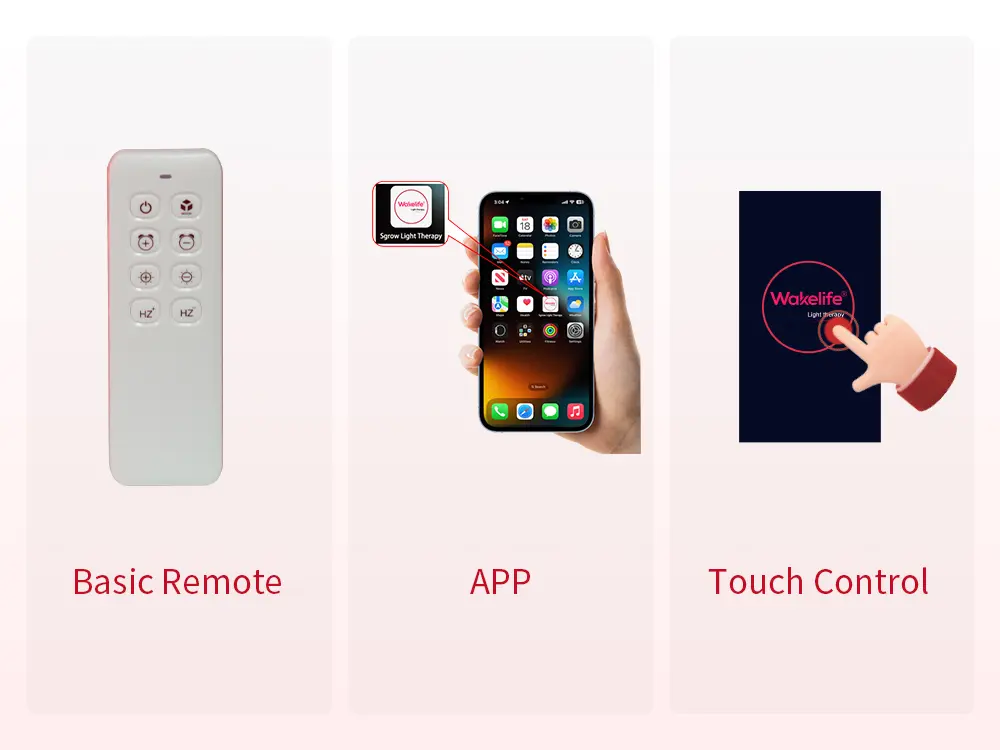
ESPLUS750 supports both APP control and touchscreen operation, com 5-escurecimento do canal, 4 modos predefinidos, 2 modos personalizados, e multi-device daisy-chain connection. For OEM/ODM brand partners, Estilo de interface do APP, mode names, and selected control functions can be adapted to match brand positioning and target market requirements.
Criterion 4 — Customization Depth
One of the most important clarifications in any sourcing conversation is the difference between private label and OEM/ODM customization. These terms are used inconsistently across suppliers, and the gap between them has a direct impact on what your brand can actually achieve.
Marca própria typically means: logo on the product, your brand name on the packaging, your language in the manual. The product itself is unchanged. You are selling the supplier’s product under your name.
OEM/ODM customization means: meaningful changes to the product itself — wavelength configuration, LED specification, lógica de controle, mode architecture, housing appearance, APP interface, and potentially product structure. The product is developed with your brand as a meaningful input, not just a label.
For brands in competitive markets, the difference matters significantly. A private label product can be copied by any other brand working with the same supplier. A genuinely customized product is harder to replicate and creates stronger long-term brand equity.
The Three Layers of Customization
Surface layer (available to most private label programs):
- Logotipo do produto
- Design de embalagem
- Instruction manual language
- Gift box and outer carton
Function layer (available to OEM/ODM programs with qualifying MOQ):
- Configuração de comprimento de onda
- LED specification and binning standard
- Dimming channel organization
- Mode settings and brightness levels
- Pulse frequency parameters
Experience layer (available to advanced OEM/ODM programs):
- APP logo and interface style
- Mode naming and user-facing language
- Brand color system in digital interface
- Boot animation and startup settings
- User guidance content within the app
Matching Customization Level to Brand Stage
New brands / first-time category entry: Start with surface-layer customization to reduce development time and MOQ requirements. Focus on selecting the right base platform rather than deep product changes.
Growth-stage brands: Invest in function-layer customization to create product specifications that competitors cannot easily replicate. Wavelength configuration and control architecture are the highest-leverage points.
Established brands: Full experience-layer customization — including APP integration — creates a connected product ecosystem that supports customer retention, usage data, and long-term brand differentiation.
Questions to Ask Your Supplier
- What is the MOQ for each level of customization?
- Which customization options require new tooling, and what is the tooling cost and lead time?
- Can the APP be white-labeled, and what elements of the interface can be modified?
- What is the development and sample timeline for a full OEM/ODM project?
- Is there a structured intake process for defining customization requirements?
The ESPLUS750 Example

ESPLUS750 supports customization across all three layers — from logo and packaging through wavelength configuration and control logic, to APP interface and mode naming. Wakelife Beauty’s OEM/ODM team works with brand partners through a structured requirement intake process to define the appropriate customization scope based on target market, quantidade do pedido, and launch timeline. MOQ varies by customization level and project scope.
Why “We Have CE and FCC” Is Not a Complete Answer
Certification is one of the most misunderstood areas in red light therapy device sourcing. Brands frequently make sourcing decisions based on a supplier’s general certification list — and later discover that the specific certifications needed for their target market, channel, or product claims are either incomplete or not applicable to the specific model they ordered.
The two most important things to understand about certifications in this category:
Primeiro: Certification scope is model-specific, not factory-wide. A factory that holds ISO 13485 certification has a quality management system that meets that standard. It does not mean every product they make automatically carries every certification. Certifications for specific product models require separate testing and documentation.
Second: Different markets require different certifications. “We’re CE and FCC certified” covers parts of the EU and US regulatory landscape — but it does not cover UKCA for the UK market, TGA or SAA for Australia, or the specific requirements of medical device registration in markets where your product may be classified differently.
Market-by-Market Certification Framework
Estados Unidos:
- FCC for electromagnetic compatibility (eletrônicos de consumo)
- FDA 510(k) clearance for devices making medical claims or classified as medical devices
- UL for electrical safety (required by some retail channels)
União Europeia:
- Marcação CE (covering LVD, Emc, and potentially MDR depending on product classification)
- RoHS for materials compliance
Reino Unido:
- UKCA (post-Brexit equivalent of CE, required for UK market access)
Austrália:
- SAA for electrical safety
- TGA registration for medical devices or devices making therapeutic claims
Canadá:
- Health Canada Medical Device License (Mdl) for regulated device categories
Global:
- ISO 13485 sistema de gestão da qualidade (required by many regulated market buyers and distributors)
- MDSAP (recognized by FDA, TGA, Saúde Canadá, e outros)
- IEC 60601 for medical electrical equipment safety (relevant for professional and clinical channels)
The Two Levels of Compliance to Evaluate
Product safety certifications — testing that verifies the device is safe for use (electrical, Emc, optical). These are typically required for market entry regardless of product claims.
Market-specific registration or clearance — additional regulatory steps required when a product makes therapeutic claims, is classified as a medical device, or is sold through medical or clinical channels. This is where many brands underestimate the complexity.
Questions to Ask Your Supplier
- Which certifications are held for the specific model I am sourcing, not the factory in general?
- Does your FDA 510(k) clearance apply to this panel, or only to specific product models?
- Can you provide documentation support for UKCA, TGA, or other market-specific requirements?
- What is your process for helping OEM/ODM customers navigate certification for their target markets?
The ESPLUS750 Example

Wakelife Beauty holds ISO 13485 and MDSAP certification for its quality management system, with product-level certifications including FDA 510(k) K250830 (for applicable models), CE, UKCA, TGA, FCC, CLIMA, Ul, IEC 60601, Rohs, and BSCI. For OEM/ODM projects, our compliance team works with brand partners to review target market requirements and prepare project-specific documentation according to the product model and intended use.
Criterion 6 — Factory Capability and Process Structure
Why the Cheapest Quote Is Rarely the Lowest-Cost Option
The final criterion is the one most commonly overlooked in an initial sourcing evaluation — and the one that has the greatest impact on long-term project success.
Factory capability is not just about production scale or price. It is about the processes that determine whether your product arrives on time, at the quality level you specified, with the documentation your market requires, and without the kinds of failures that create warranty returns, distributor disputes, and brand damage.
In two earlier articles, we documented the most common OEM project failures in LED therapy device manufacturing — and the development process we use to prevent them. The consistent finding: failures do not start on the production line. They start in early-stage decisions about requirements, validação, and change management.
The same logic applies to supplier selection. A factory that skips structured validation, lacks cross-functional review processes, or has no formal change order system will eventually create problems for your brand — regardless of how good the initial sample looks.
What to Evaluate Beyond Price and Sample Quality
Validation process: Does the factory run EVT (Teste de validação de engenharia), TVP (Teste de validação de projeto), and PVT (Teste de validação de produção) as formal, documented stages? Each answers a different question: Isso pode funcionar? Será que vai durar? Pode escalar? Skipping any of these transfers unresolved risk to your mass production run.
Cross-functional review: Is there a structured process for involving R&D, cadeia de mantimentos, qualidade, and production teams in project evaluation — or does one person make all the calls? Single-point ownership is a risk model in complex device development.
Change management: What happens when you need to change a specification after tooling has started? A factory with a formal Engineering Change Order (ECO) process can handle changes systematically, with documented cost and timeline impact. A factory without one absorbs changes informally — and the costs appear later, often unexpectedly.
Production capacity and stability: Can the factory handle your current order volume and your projected growth? Is the supply chain stable enough to maintain consistent component quality across production runs?
Quality system certification: ISO 13485 certification indicates a quality management system that meets international standards for medical device manufacturing. For brands targeting regulated markets, working with an ISO 13485-certified supplier significantly simplifies compliance documentation and distributor qualification.
Questions to Ask Your Supplier
- Do you run EVT, TVP, and PVT for new product development programs? Can you share an example of a DVT summary report?
- What does your cross-functional project review process look like?
- How do you handle change requests after production authorization?
- What is your monthly production capacity, and how is capacity allocated across customers?
- What quality certifications does your manufacturing system hold?
The Wakelife Beauty Example
Wakelife Beauty’s manufacturing facility in Shenzhen covers 20,000㎡ with 12 linhas de produção e uma capacidade mensal de 180,000+ unidades. Our development process runs mandatory EVT, TVP, and PVT stages for new product programs, with cross-functional review involving R&D, design de produto, cadeia de mantimentos, and quality teams from project initiation. All post-authorization changes follow a formal ECO process with documented cost and timeline impact before work resumes. Our quality management system is certified to ISO 13485 e MDSAP.
For a detailed explanation of how this process works in practice — and the real failures that made each step necessary — see our companion article: Processo de desenvolvimento de dispositivo de terapia de luz vermelha: Lições das falhas do mundo real
How to Use These 6 Criteria to Build Your Shortlist
The six criteria above give you a framework for evaluating suppliers beyond price and LED count. The table below translates each criterion into a specific question and a verification method you can use in supplier conversations.
| Criterion | Key Question | Como verificar |
|---|---|---|
| Configuração de comprimento de onda | Does the panel go beyond 660nm + 850nm? What binning tolerance? | Request full wavelength spec and binning standard documentation |
| Irradiance data | Are multi-distance measurements available? | Request irradiance data at 3", 9", e 12" with test methodology |
| Control system | Does it support APP, escurecimento, and custom modes? | Request product demo or video; ask about APP customization scope |
| Customization depth | What can actually be changed, at what MOQ? | Request customization option list with MOQ and lead time by level |
| Certificações | Which certs are held for this specific model and market? | Request model-specific cert documentation for your target market |
| Factory and process | Are EVT/DVT/PVT run? Is there a formal ECO process? | Request process overview and example validation documentation |
Matching Criteria Priority to Brand Stage
Not every criterion carries equal weight at every stage of brand development. Here is a simplified framework:
New brands entering the red light therapy category for the first time: Prioritize customization depth and MOQ flexibility. You need a supplier who can support a smaller initial program without requiring deep product development investment. Surface-layer customization with a strong base platform is the right starting point.
Growth-stage brands expanding an existing product line: Prioritize wavelength configuration and control system. These are the two dimensions that create the most visible product differentiation and support premium pricing. A brand that has already established market presence can invest in function-layer customization to separate its products from the competition.
Established brands managing a multi-SKU portfolio in regulated markets: Prioritize certification coverage and factory process structure. At this stage, compliance gaps and production inconsistencies create the most serious business risk. A supplier with ISO 13485 certificação, structured validation processes, and documented change management is a materially lower-risk partner than one without these systems — regardless of price.
Perguntas frequentes
What wavelengths should a red light therapy panel have for brand differentiation?
No mínimo, a red light therapy panel should include 660nm red light and 850nm near-infrared. For stronger brand differentiation, intermediate configurations add 630nm, 810nm, and 830nm. Advanced panels include 1060nm deep near-infrared, which creates a genuine technical separation from standard panels and supports premium product positioning in wellness and professional markets.
What is the difference between private label and OEM/ODM for red light therapy panels?
Private label means adding your logo, embalagem, and manual to an existing product without changing the product itself. OEM/ODM customization means meaningful changes to the product — wavelength configuration, LED specification, lógica de controle, APP interface, and housing appearance. Private label products can be replicated by any brand working with the same supplier. Genuinely customized OEM/ODM products create stronger long-term brand equity.
What certifications does a red light therapy panel need for different markets?
Certification requirements vary by market. For the United States: FCC for EMC, UL for electrical safety, FDA 510(k) for medical device claims. For the EU: CE marking and RoHS. For the UK: UKCA. For Australia: SAA for electrical safety and TGA for therapeutic devices. ISO 13485 quality management certification is recommended globally. Importantly, certifications are model-specific — confirm which certifications apply to the exact model you are sourcing, not just the factory in general.
What factory capability should I evaluate when choosing a red light therapy panel supplier?
Beyond price and sample quality, evaluate whether the factory runs formal EVT (Teste de validação de engenharia), TVP (Teste de validação de projeto), and PVT (Teste de validação de produção) stages. Ask about their cross-functional project review process, how they handle change requests after production authorization, their monthly production capacity, and whether their quality management system is ISO 13485 certificado.
How should a new brand prioritize criteria when choosing a red light therapy panel?
New brands should prioritize customization depth and MOQ flexibility — start with surface-layer customization and a strong base platform. Growth-stage brands should prioritize wavelength configuration and control system to create differentiation. Established brands in regulated markets should prioritize certification coverage and factory process structure, as compliance gaps and production inconsistencies create the most serious business risk at that stage.
Conclusão: Choose a Platform, Not Just a Panel
The red light therapy panel market will continue to grow. It will also continue to become more competitive, more commoditized at the entry level, and more demanding at the premium end — as brands, distribuidores, and professional buyers become more sophisticated in their sourcing criteria.
For brands building a serious product line, the sourcing decision is not just about finding a panel that meets today’s specification at today’s price. It is about choosing a product platform and a manufacturing partner that can support the brand’s growth over time — through product iteration, market expansion, regulatory evolution, and increasing customer expectations.
The six criteria in this article — wavelength configuration, irradiance transparency, control system, profundidade de personalização, certification coverage, and factory process structure — are the dimensions that determine whether a supplier can actually deliver on that kind of long-term partnership.
ESPLUS750 is one example of a panel platform designed with these criteria in mind: six wavelengths including 1060nm deep NIR, multi-distance irradiance data, APP control and touchscreen operation, 5-escurecimento do canal, OEM/ODM customization across all three layers, and manufactured under ISO 13485 and MDSAP-certified quality systems with structured EVT/DVT/PVT validation.
But the criteria apply regardless of which panel you ultimately choose. Use them to ask better questions, surface hidden risks earlier, and make a sourcing decision that your brand will still be comfortable with twelve months after the first order.
Related Reading
- Processo de desenvolvimento de dispositivo de terapia de luz vermelha: Lições das falhas do mundo real — The full 7-stage OEM development process behind consistent product quality
- 5 Falhas reais de OEM em dispositivos de terapia LED – e como evitá-las — Real cases where sourcing decisions created downstream problems
- 12 Fabricantes de máscaras faciais LED em Shenzhen: A Sourcing Guide — Evaluated supplier profiles for the LED face mask category
Ready to Evaluate ESPLUS750 for Your Brand?
If you are sourcing a red light therapy panel and want to apply the six criteria above to a specific project, Wakelife Beauty’s OEM/ODM team can help you:
- Confirm which ESPLUS configuration matches your target market and positioning
- Review customization options and MOQ requirements for your program scope
- Identify certification requirements for your target market
- Request samples and a project-specific quotation
To help us give you the most useful response, share the following when you reach out:
- Target market and sales channel
- Estimated order quantity
- Customization requirements
- Certification or compliance needs
- Expected launch timeline
👉 View ESPLUS750 full specifications: https://wakelifebeauty.com/product/esplus750-red-light-therapy-panel/
👉 Explore our OEM/ODM services: https://wakelifebeauty.com/oem-odm/
👉 Revise nossas credenciais de qualidade e conformidade: https://wakelifebeauty.com/quality-compliance/








