Qualità & Conformità
Structured systems, validated outcomes, and defined accountability — across every stage of manufacturing.
Audit-Ready Execution: Beyond Marketing Claims
The Daily Evidence Chain of Medical QMS Compliance
In the medical device manufacturing industry, the gap between marketing claims and actual factory execution is often exposed during on-site audits.
At WAKELIFE, compliance is not a certificate on the wall—it is an unbreakable chain of daily evidence.
Cross-Functional Quality Culture
Quality is a company-wide mandate, not just a QA department task.
Quality responsibility at WAKELIFE extends beyond the QC department. From engineering to the assembly line, every team member strictly adheres to Standard Operating Procedures (SOPs). This cross-functional accountability ensures quality is built into the product, not just inspected at the end.
The Unbreakable Evidence Chain
Real daily execution logs, not overnight audit preparations.
True quality management cannot be fabricated overnight. We rely on real-time ERP data and a closed-loop tracking system. Every production log, CAPA report, and batch record is continuously accumulated, making our daily execution impossible to fake.
Medical-Grade Execution Baseline
Exceeding standard requirements with strict ISO 13485 conformità.
We apply ISO 13485 medical device quality standards to all production lines, regardless of the product tier. By maintaining this medical-grade baseline, we guarantee 100% alignment with our OEM/ODM clients’ most stringent QA requirements.
Always Audit-Ready
We invite unannounced on-site validations from professional clients.
Because our documentation reflects actual daily operations rather than rushed audit preparations, our facility remains in an “Audit-Ready” state every single day. We welcome QA directors and compliance officers to verify our manufacturing excellence in person.
WAKELIFE’s manufacturing operations are governed by a structured ISO 13485–aligned quality management system, ensuring that production activities are executed within defined procedures rather than individual discretion.
This system establishes how processes are documented, monitored, and continuously improved across the manufacturing lifecycle—supporting consistency, tracciabilità, and regulatory readiness for medical-grade red light therapy devices.
Quality requirements are embedded into operational workflows, enabling manufacturing outcomes to be guided by standardized controls rather than ad-hoc adjustments.
- Oversight mechanisms ensure system implementation alignment.
- Medical-Grade QMS: Iso 13485 & MDSAP Certified Facility
01 — The Framework
Sistema di gestione della qualità
Production activities are executed within defined procedures rather than individual discretion.
Medical-Grade Manufacturing for Premium Wellness Devices
Proudly ISO 13485 & MDSAP Certified.
We manufacture your red light therapy masks and panels to the world’s strictest medical device quality standards — so you can sell with confidence in every market.
One Audit. Five Countries. Zero Compromise.
Our facility holds MDSAP (Medical Device Single Audit Program) certification — the highest level of manufacturing compliance recognition in the global medical device industry.

| Regulatory Body | Paese |
|---|---|
| FDA (Food & Drug Administration) | Stati Uniti |
| Health Canada | Canada |
| ANVISA | Brazil |
| MHLW / PMDA | Japan |
| TGA (Amministrazione delle merci terapeutiche) | Australia |
Zero compliance risk
your products are backed by manufacturing infrastructure already accepted by the world's most stringent regulatory bodies.
Future-proof supply chain
whether you're launching a wellness brand today or seeking FDA/CE medical device clearance tomorrow, our factory is ready.
Full traceability
every component, every batch, every shipment is documented and traceable end-to-end.
WAKELIFE’s quality management system is supported by defined verification and testing capabilities that ensure manufacturing outcomes are validated rather than assumed.
In-House Environment
Our in-house testing environment enables performance consistency, functional stability, and production readiness to be assessed throughout the manufacturing process.
Integrated Workflows
Verification activities are integrated into production workflows, allowing quality requirements to be evaluated continuously—from component integration through final assembly.
"This structured approach ensures that manufacturing decisions are informed by measurable validation rather than subjective judgment."
02 — The Validation
Verification & Testing Capability
We don’t just assemble devices; we validate them. Our core equipment testing ensures that every unit bearing your brand name meets strict medical-grade standards.
Validated Outcomes
Not Assumed Performance

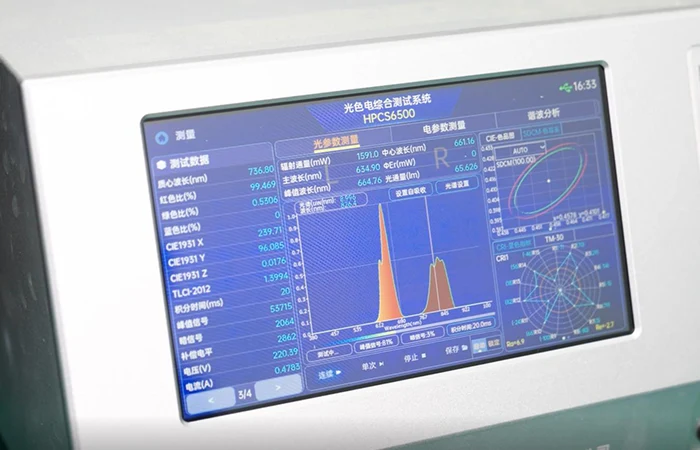
Integrating Sphere
Spectral Analysis System
Technical Standard
Verifies emitted wavelength strictly within a ±5nm tolerance. We test every batch, never just relying on LED datasheets.
Eliminates wavelength uncertainty. Guarantees your end-users receive authentic, medical-grade light therapy rather than standard LED illumination, elevating your brand’s clinical credibility.
Climate Chamber
Extreme Environment Simulation
Test Parameters
Range: -20°C to +60°C
Humidity: 95% RH
Ensures flawless operation across diverse global climates. Whether shipping to cold Nordic regions or humid Southeast Asia, internal components remain protected against environmental stress.

Drop Tester
Structural Integrity Test
Impact Specs
Height: 1.2M
Metodo: 6 angles, marble floor impact.
Simulates real-world accidental drops to ensure the housing remains intact and internal circuits stay connected. This directly minimizes after-sales returns (RMA) and protects your brand reputation.

Salt Spray Tester
Corrosion Resistance Test
Exposure Time
24h – 48h continuous salt fog exposure.
Specifically tests against facial sweat and cosmetic residue. Prevents oxidation and poor contact on charging pins and metal parts, significantly extending the product’s lifespan.

Battery Aging Cabinet
Cycle Life Testing
Performance Goal
Capacità > 80% Dopo 500 Cicli di carica.
Prevents premature battery degradation. Sustaining high capacity after hundreds of uses ensures a premium, long-lasting experience that aligns with high-end beauty brand standards.
- Not What We Say
“Advanced testing equipment”
“Large-scale laboratory”
“State-of-the-art facilities”
- What It Means
Outcomes are validated, not assumed
Decisions are measurable, not subjective
The system is actually executed
Quality is the Best Business Plan
We invite all our OEM/ODM partners to visit our Shenzhen facility and witness these rigorous
Iso 13485 testing procedures in person.
03 — The Recognition
Regulatory Certifications
WAKELIFE’s regulatory certifications reflect the practical implementation of its quality management system across manufacturing operations.
“These certifications are not standalone achievements but indicators of structured quality practices and validated manufacturing discipline.”
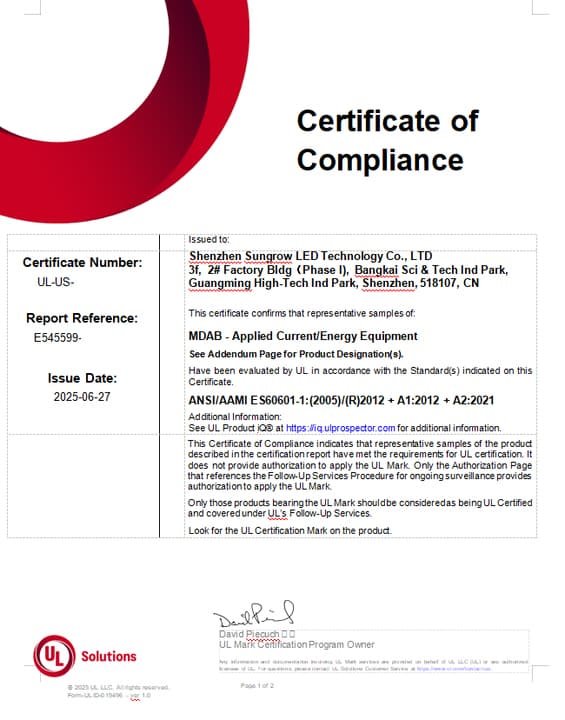
FDA NO. (3033883954)
Certificati emessi dagli Stati Uniti. Food and Drug Administration è disponibile su Sito web FDA ufficiale.
Iso 13485 / MDSAP
Iso 13485 è uno standard di sistema di gestione della qualità da utilizzare nell'ambiente di regolamentazione dei dispositivi medici.
Ul
Independent safety certification supporting electrical safety compliance and trusted market acceptance.
Mdl
La licenza del dispositivo medico canadese (Mdl) è equivalente agli Stati Uniti. FDA 510(k) spazio.
FCC
La certificazione FCC è una certificazione obbligatoria di prodotti elettronici negli Stati Uniti.
BSCI
La certificazione BSCI è una certificazione Social Social Business progettata per garantire che le prestazioni di responsabilità sociale di un'azienda nelle catene di approvvigionamento globali soddisfino gli standard internazionali.
Rohs
La certificazione ROHS è uno standard obbligatorio stabilito dalla legislazione dell'Unione Europea. Il suo nome completo è il “Limitazione della direttiva di sostanze pericolose.”
UKCA
UK conformity marking supporting compliance with applicable UK product safety and regulatory requirements.
ISO9001
ISO9001 si riferisce a uno standard di certificazione del sistema di gestione della qualità sviluppato dall'Organizzazione internazionale per la standardizzazione (Iso).
ISO14001
La certificazione del sistema di gestione ambientale ISO4001 è uno standard di sistema di gestione ambientale rilasciato dall'organizzazione internazionale per la standardizzazione (Iso).
ISO45001
Iso 45001 è uno standard del sistema di gestione della salute e della sicurezza sul lavoro pubblicato dall'Organizzazione internazionale per la standardizzazione (Iso).
04 — The Accountability
Governo della qualità & Leadership
"This structured approach ensures that manufacturing decisions are informed by measurable validation rather than subjective judgment."
Responsibilities for quality management and compliance are clearly assigned, enabling system implementation to be monitored, maintained, and continuously aligned with regulatory expectations.
This governance approach reflects how regulated manufacturing environments operate—where quality outcomes are shaped not only by processes, but by structured leadership and responsibility.
The Complet Quality Logic
Who Oversees the System?
Learn how quality and compliance responsibilities are defined and overseen across WAKELIFE’s manufacturing operations.