Introduction: The Growing Demand for Wearable Light Therapy
The global red light therapy market is experiencing unprecedented growth. Consumers are no longer satisfied with stationary panels or bulky clinical devices — they want portable, hands-free, wearable solutions that fit seamlessly into their daily routines.
Si’s a fitness enthusiast recovering from an intense workout, an office worker dealing with chronic lower back discomfort, or a wellness brand looking for the next high-margin product to launch — the red light therapy belt has emerged as one of the most in-demand product categories heading into 2026.
But not all therapy belts are created equal. Most products on the market use basic fabric construction, single-wavelength LEDs, and wired controllers — resulting in mediocre user experiences and limited brand differentiation.
Que’s exactly why Wakelife engineered the BE-G122 Red Light Therapy Silicone Belt — a next-generation wearable device designed to set a new standard in both performance and user experience.
What Is the Wakelife BE-G122?
The BE-G122 is a 2-in-1 red light and vibration therapy belt built with food-grade standard silicone, featuring a wireless magnetic snap-on controller et 122 triple-chip (Triple-Chip) LEDS delivering clinically relevant irradiance at the skin surface.
Unlike traditional therapy belts made from fabric or neoprene, the BE-G122 uses food-grade standard silicone — a material trusted in skincare and food-contact applications — making it sweat-resistant, odor-free, and incredibly easy to clean.
Spécifications clés en un coup d'œil
| Paramètre | Spécification |
|---|---|
| Modèle | BE-G122 |
| Matériel | Silicone |
| Quantité LED | 122 PCS (Triple-Chip 5050) |
| Total des jetons | 122 × 3 = 366 puces |
| Longueur d'onde | 660nm (Rouge) + 850nm (Nir) |
| Irradiance | 30 mW/cm² à la surface de la peau |
| Vibration | 4 moteurs, 3 niveaux d'intensité |
| Batterie | 2,000Lithium-ion mAh |
| Chargement | USB Type-C, ~3.5 hours |
| Minuteur | 10 mon arrêt automatique |
| Conformité | CE / FCC / Rohs |
5 Features That Make the BE-G122 Stand Out
1. High-Energy Triple-Chip Dual-Wavelength Technology
The BE-G122 uses 122 triple-chip (Triple-Chip) 5050 LEDS, with each LED housing 3 jetons individuels — totaling 366 light-emitting chips across the entire belt.


This triple-chip architecture delivers two therapeutic wavelengths simultaneously:
- 660feu rouge — Targets the epidermis and dermis layers, promoting collagen synthesis, improving blood circulation, and supporting skin cell regeneration.
- 850nm Near-Infrared (Nir) Lumière — Penetrates deeper into subcutaneous tissue and muscle, stimulating mitochondrial ATP production, Réduire l'inflammation, and accelerating tissue repair.
Two selectable modes:
- Mode 1: 660nm only — for surface-level skin and circulation benefits
- Mode 2: 660nm + 850nm combined — for deep tissue penetration and recovery
The measured irradiance of 30 MW / CM² at the skin surface places the BE-G122 firmly in the professional-grade category, far above the sub-10 mW/cm² output of many consumer-grade alternatives.

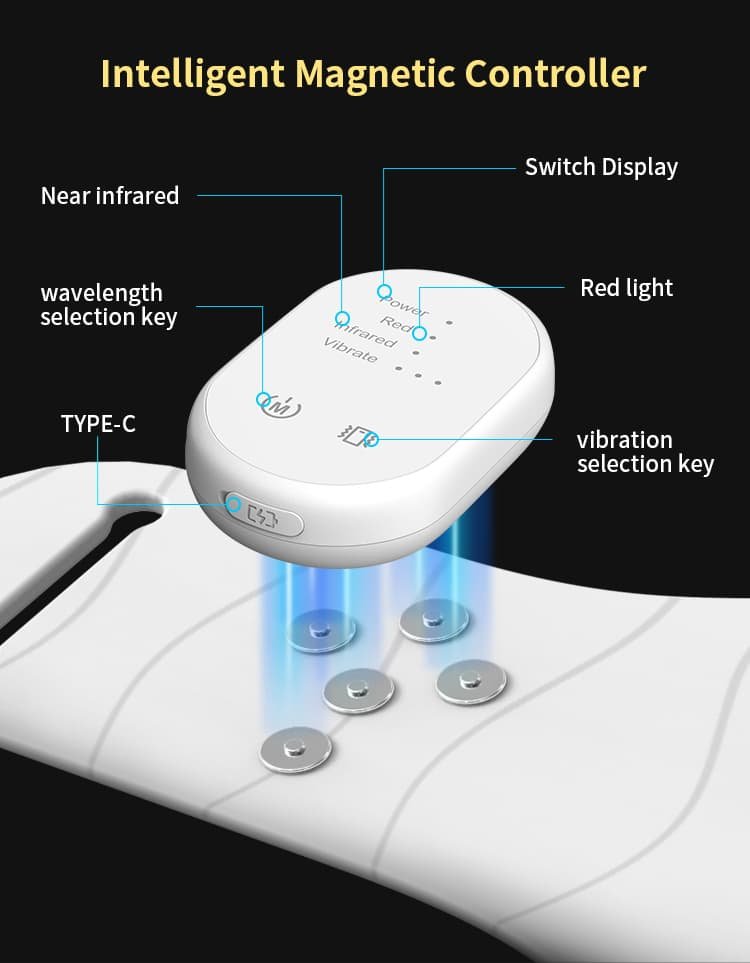
2. Wireless Magnetic Snap-On Controller
One of the most innovative features of the BE-G122 is its contrôleur magnétique amovible. Unlike traditional belts that rely on wired control boxes or built-in buttons that are hard to reach, the BE-G122’s controller snaps on and off with a simple magnetic connection — no tools, no cables, no hassle.
This design delivers three key advantages:
- Premium User Experience — The satisfying magnetic « click » and tool-free operation create a tactile, high-end feel that justifies premium retail pricing.
- Easy Cleaning — Detach the controller, wipe down the silicone belt, and reattach. No electronic components to worry about during cleaning.
- Reliable Connection — The magnetic interface provides a stable, secure connection during movement — whether the user is resting, faire de l'exercice, or doing household chores.
The controller features clear LED indicators for Power, Feu rouge, Infrarouge, and Vibration status, along with a USB Type-C charging port for universal compatibility.
3. Built-In 4-Motor Vibration Massage System
The BE-G122 goes beyond light therapy alone. It integrates four vibration motors avec three adjustable intensity levels, creating a synergistic combination of photobiomodulation and mechanical massage.
Ce 2-in-1 approach offers users:
- Enhanced blood circulation in the treatment area
- Improved lymphatic drainage
- More effective delivery of light energy through micro-vibration
- A more engaging, spa-like user experience
Pour les marques, this dual-function capability creates a stronger value proposition and more compelling marketing story compared to light-only or vibration-only devices.

4. Food-Grade Standard Silicone Construction
Material choice is critical in wearable wellness devices. The BE-G122 uses food-grade standard silicone — a deliberate upgrade from the fabric, neoprene, or basic TPU materials commonly found in competing products.
Why Food-Grade Silicone Matters
| Fonctionnalité | Fabric / Neoprene Belts | BE-G122 Food-Grade Silicone |
|---|---|---|
| Sweat absorption | Absorbs & retains odor | 100% essuyer |
| Skin safety | Varies by material | Food-grade standard |
| Durabilité | Degrades over time | Long-lasting, flexible |
| Cleaning | Requires washing / drying | Simple wipe-down |
| Aesthetic | Looks worn after use | Maintains premium look |
For comparison, many competitors still use OK Cloth + TPU construction, which absorbs sweat and becomes difficult to maintain over time.

5. Versatile Multi-Area Application
The BE-G122 is designed with an adjustable elastic strap, making it suitable for multiple body areas:
- Lower back — The most common use case for pain relief and muscle recovery
- Abdomen — Popular among postpartum recovery and wellness consumers
- Upper back & épaules — For users with desk-related tension
- Thighs & knees — Athletes and active users seeking targeted recovery
This versatility allows brands to market a single SKU across multiple consumer segments — maximizing product reach and ROI.
Who Is This Product For?
Pour les marques & Distributeurs (B2B)
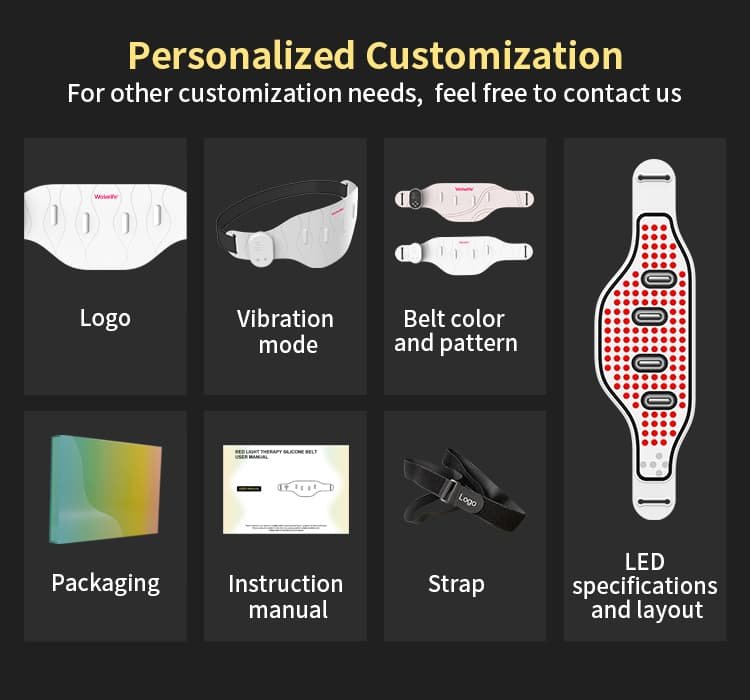
The BE-G122 is built as a private-label-ready OEM/ODM platform. Wakelife supports full customization including:
Options de personnalisation
| Option de personnalisation | Détails | MOQ |
|---|---|---|
| Impression de logos | Silk-screen printing on belt or controller | 100 PCS |
| Emballage personnalisé | Coffret cadeau, boîte à couvercle magnétique, Insertion EVA | 200 PCS |
| Sélection de la longueur d'onde | 415 / 460 / 590 / 630 / 660 / 830 / 850 nm | 500 PCS |
| Couleur du silicone | Pantone color matching available | 200 PCS |
| Développement ODM complet | Custom industrial design, structure, and feature development | Contactez-nous |
* Flexible OEM/ODM solutions available — from private label to fully customized product development.
Wakelife’s parent company, Technologie Cie. de Shenzhen Sungrow LED., Ltée, was founded in 2015 and has grown through continuous expansion — from early red light therapy focus to becoming an Alibaba SKA TOP1 Supplier by 2020, expanding its factory to 10,000m²+ by 2023, and achieving MDSAP and ISO 13485 certification by 2024.
Dans 2025, the company secured FDA 510(K) autorisation for its LED device platform, with plans to expand its R&D team from 40 à 50+ engineers by 2026 and pursue additional medical certifications for 10+ products.
For End Consumers (B2C Context)
For the end user, the BE-G122 delivers a spa-quality, hands-free therapy experience that integrates effortlessly into daily life:
- 📱 While resting — Relax on the couch with a 10-minute therapy session
- 😴 While sleeping — The auto-off timer ensures safe, worry-free use
- 🏋️ While exercising — The wireless design allows full freedom of movement
- 🏠 While doing housework — Light therapy doesn’t require dedicated time
Each full charge supports jusqu'à 12 séances (10 minutes each), providing over 120 minutes of total therapy time before recharging.
How the BE-G122 Compares to Other Therapy Belts
Product Comparison
| Fonctionnalité | Generic Fabric Belt | Standard Belt (e.g. BE-120) | Wakelife BE-G122 |
|---|---|---|---|
| Matériel | Neoprene / Fabric | OK Cloth + TPU | Silicone de qualité alimentaire |
| LED Architecture | Single-chip | Triple-chip | Triple-chip (366 jetons au total) |
| Longueurs d'onde | 660 nm seulement | 660 nm + 850 nm | 660 nm + 850 nm (customizable) |
| Irradiance | 5–15 mW/cm² | 15–25 mW/cm² | 30 MW / CM² |
| Contrôleur | Câblé | Built-in or wired controller | Wireless magnetic snap-on controller |
| Vibration | Aucun | Basic vibration | 4 motors with 3 niveaux d'intensité |
| Cleaning | Difficult to clean | Moderate maintenance required | Easy wipe-clean surface |
| Certifications | Often none | CE | CE + FCC + Rohs |
* BE-G122 is designed under a medical-grade manufacturing system to ensure consistency, sécurité, et performances.
The BE-120 and other belt models in Wakelife’s lineup use OK Cloth + TPU construction, while the BE-G122 represents the premium silicone tier — designed for brands targeting higher price points and more discerning consumers.
The Science Behind Red Light Therapy Belts

Thérapie par lumière rouge, also known as photobiomodulation (PBM), works by delivering specific wavelengths of light energy to biological tissue. When cells absorb this light — particularly at 660nm and 850nm — it triggers a cascade of beneficial cellular processes:
Mitochondrial Activation — Light photons are absorbed by cytochrome c oxidase in the mitochondrial electron transport chain, increasing ATP production — the cell’s primary energy currency.
Increased Blood Flow — Nitric oxide is released, causing vasodilation and improved microcirculation in the treated area.
Inflammation réduite — Pro-inflammatory cytokines are downregulated while anti-inflammatory mediators are upregulated.
Accelerated Tissue Repair — Enhanced cellular energy and improved nutrient delivery support faster healing of muscles, tendons, and connective tissue.
The BE-G122’s combination of 660nm + 850nm at 30 MW / CM² with a 10-minute treatment protocol delivers a light dose of approximately 18 J/cm² — well within the therapeutic window identified in peer-reviewed research.
Qu'y a-t-il dans la boîte?
Every BE-G122 unit ships with a complete kit:
- ✅ Main Device × 1 — The food-grade silicone red light therapy belt
- ✅ Wireless Magnetic Controller × 1 — Snap-on control unit
- ✅ USB-A to Type-C Charging Cable × 1
- ✅ Adjustable Elastic Strap × 1 — Fits up to 50″ waist
- ✅ Portable Storage Bag × 1
- ✅ User Manual × 1
For OEM partners, all components — including packaging, manuel, strap design, and storage bag — can be fully customized to match your brand identity.
Manufacturing Excellence Behind the BE-G122
Wakelife’s manufacturing journey began in 2015 with a focus on red light therapy. Key milestones include:
Development Milestones
| Année | Jalon |
|---|---|
| 2015 | Shenzhen Sungrow founded, focusing on red light therapy |
| 2018 | Manufacturing facility established |
| 2020 | Recognized as Alibaba SKA TOP1 Supplier |
| 2021 | Factory expanded to 4,200 m² |
| 2022 | Initiated R&D in beauty device category |
| 2023 | Factory expanded to 10,000 m² |
| 2024 | Atteint MDSAP et ISO 13485 certification |
| 2025 | FDA 510(k) clearance obtained |
| 2026 | R&D team expanding to 50+ ingénieurs; 10+ products undergoing medical certification |
* Continuous investment in R&D and compliance ensures long-term product reliability and global market readiness.
Every BE-G122 unit undergoes rigorous quality verification:
- Wavelength Testing — Integrating sphere spectral analysis confirms ±5nm accuracy
- Irradiance Verification — Consistent 30 mW/cm² output across batches
- Environmental Stress Testing — Climate chamber simulation from -20°C to +60°C
- Drop Testing — 1.2m, 6-angle impact tests
- Battery Cycle Testing — Capacity >80% après 500 cycles de charge
This testing discipline reflects Wakelife’s ISO 13485 quality management system.
How to Get Started with the BE-G122
Next Steps:
- Demander un échantillon — Test the product quality firsthand
- Discuss Customization — Logo, couleur, conditionnement, wavelength preferences
- Review Compliance Documentation — CE, FCC, RoHS certificates and ISO 13485 audit reports
- Plan Your Launch — From sample to mass production
Intéressé par le commerce de gros, marque personnalisée, ou distribution?
Questions fréquemment posées
Quelle est la quantité de commande minimale?
Standard wholesale MOQ is 10 pièces. Custom logo starts at 100 pièces. Full customization starts at 200–500 pieces.
Can I customize the wavelength?
Oui. Standard is 660nm + 850nm. Custom wavelengths available across 415nm, 460nm, 590nm, 630nm, 660nm, 830nm, et 850 nm (MOQ 500 PCS).
How long does each treatment session last?
10 minutes with auto-off timer. Jusqu'à 12 séances per full charge
What certifications does the BE-G122 have?
CE, FCC, et Rohs. The facility is ISO 13485 and MDSAP certified, and Wakelife holds FDA 510(K) autorisation (K250830).
What warranty is provided?
12-month warranty covering manufacturing defects. Extended warranty available for OEM partners.
How does the magnetic controller work?
The controller attaches via built-in magnetic connection points — align and snap on. Detach easily for cleaning or charging.
Ready to bring the BE-G122 to your brand? Get in touch with our team to start your project today.