ODM / OEM
- Monatliche Produktion von 180.000
- Passen Sie die perfekte LED -Schönheitsmaske für Ihre Marke so schnell an wie an 7 Tage.

Erwecken Sie Ihre Vision zum Leben
Nutzen Sie unseren neuesten R-R&D und Design -Expertise, um Ihre einzigartigen Ideen in die Realität umzusetzen. Teilen Sie Ihr Konzept, und wir liefern eine maßgeschneiderte Lösung, die auf Ihre Anforderungen zugeschnitten ist.

Nahtlose Anpassung, Beginnen Sie zu Ende
Unser dedizierter r&D, Maschinenbau, und Designteams arbeiten eng mit Ihnen zusammen, um das perfekte LED -Schönheitsgerät zu erstellen. Vom ersten Konzept zum Endprodukt, Wir sorgen für eine glatte, effizienter Prozess mit schnellen Abwicklungszeiten.

Massenproduktion mit Effizienz
Mit einem Team von 150+ qualifizierte Fachkräfte und hochmoderne automatisierte Produktionslinien, Wir zeichnen uns im Umgang mit groß angelegten Bestellungen aus. Unsere optimierten Prozesse sorgen für eine rechtzeitige Lieferung, Helfen Sie, Ihre Produkte schneller auf den Markt zu bringen.

24/7 Zuverlässige Unterstützung
Unser Kundendienstteam ist rund um die Uhr verfügbar, um alle Anforderungen nach dem Verkauf zu befriedigen, Gewährleistung eines nahtlosen und stressfreien Erlebnisses für Sie und Ihre Kunden.
Was können wir anpassen??
Unsere Masken helfen Ihnen dabei, eine tiefe Verbindung mit den Verbrauchern aufzubauen und eine vertrauenswürdige Schönheitsmarke zu schaffen.

Erscheinungsanpassung
- Muster
- Form
- LOGO
- Bedienungsanleitung
- Farbe
- Verpackung
Interne Komponenten
- LED -Chipauswahl
- LED -Perlenmenge und -anordnung
- FPC -Board
Controller -Anpassung
- App -Anpassung
- Controller -Griff
Controller -Anpassung
- Bestrahlung: 10-50MW/C㎡
- Wellenlänge: 300NM-2000nm
Unsere OEM/ODM -Produktpalette
Unsere Zertifizierung von Drittanbietern
Wir halten alle notwendigen Drittanbieterzertifizierungen, einschließlich FDA (No3033883954), FDA(510K), Ul, Mdl, MDSAP, IEC60601, ISO13485, ISO9001, ISO14001, ISO45001, BSCI, Rohs, Und was. Sie können unsere LED -Schönheitsmasken in jedem Internet oder Offline -Shop verkaufen.
LED Beauty Device OEM/ODM -Produktionsprozess
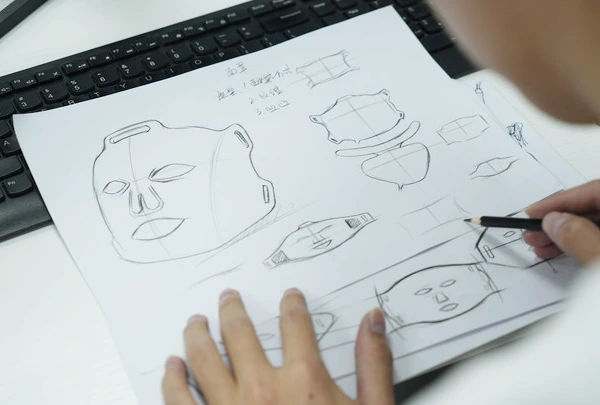
Aussehensdesign
Einschließlich Muster, Formen, Logos, Farben

Strukturelles Design
Interne Komponenten wie LED -Perlenchips, Anzahl der Perlen, FPC -Boards, usw.

3D Modelldruck
Schnelle Erzeugung von Probenmodellen durch 3D -Drucker

Etikett, Handbuch, und Verpackungsdesign
Entwerfen Sie Ihre eigenen Markenetiketten, Verpackung und Handbücher.

Produktionsstart
Nach Abschluss der ersten Vorbereitungen, Massenproduktion beginnt.

Testen
Die erste Produktcharge wird getestet, um sicherzustellen 100% Einhaltung der Qualitätsstandards.

Qualitätsinspektion
Sobald die Massenproduktion abgeschlossen ist, Jedes Produkt erfährt eine gründliche Qualitätsinspektion.

Logistik & Versand
Produkte werden über DHL versendet, UPS, FedEx, Tnt, EMS, und andere zuverlässige Träger.
Häufig gestellte Fragen zur Entwicklung von OEM/ODM-LED-Schönheitsgeräten
Was ist der Unterschied zwischen OEM und ODM?, und wie schneiden sie im Vergleich zu anderen Wakelife-Diensten ab??
OEM (Herstellung von Erstausrüstung) bedeutet, dass Sie das Produktdesign oder die Spezifikationen bereitstellen, und wir fertigen es für Sie. ODM (Original-Design-Herstellung) Das bedeutet, dass wir die gesamte Entwicklung übernehmen – vom Konzept über das Industriedesign bis hin zum Engineering, Prototyping, und Massenproduktion – basierend auf Ihrer Idee oder Ihrem Auftrag.
Hier sehen Sie den Vergleich aller vier Dienste:
- Großhandel — Kaufen Sie unsere bestehenden Produkte in großen Mengen zu wettbewerbsfähigen Preisen. Keine Anpassung erforderlich.
- Handelsmarktlabel — Fügen Sie Ihr Logo hinzu, Verpackung, und Branding unserer bestehenden Produkte. Das Produkt selbst bleibt unverändert.
- Anpassung — Spezifische Parameter ändern (Wellenlänge, Leistung, Behandlungsmodi, Farben) auf einer bestehenden Produktplattform.
- OEM / ODM — Entwickeln Sie ein völlig neues oder von Grund auf erheblich verändertes Produkt, mit vollem Engineering, Werkzeuge, und Compliance-Unterstützung.
Wenn Sie sich nicht sicher sind, welcher Weg zu Ihrem Unternehmen passt, Kontaktieren Sie unser Team und wir empfehlen Ihnen den besten Ansatz basierend auf Ihren Zielen, Budget, und Zeitleiste.
Wenn ich nur eine Idee hätte, Kann Wakelife das Produkt als vollständiges ODM-Projekt entwickeln??
Absolut. Wir bieten eine durchgängige ODM-Entwicklung – von der Produktdefinition und dem Industriedesign bis hin zum optischen/elektronischen Engineering, Prototyping, Validierung, Werkzeuge, und Massenproduktion. Für den Einstieg benötigen Sie keine technischen Zeichnungen oder Konstruktionsdateien.
Unser Team passt das Design an Ihre Zielgruppe an, Markenpositionierung, und Kostenziele bei gleichzeitiger Sicherstellung der Herstellbarkeit und Compliance-Bereitschaft. Der Prozess beginnt mit einem Treffen, um Ihr Konzept zu verstehen, Anschließend liefern wir einen klaren Vorschlag mit einem detaillierten Zeitplan.
Ob Sie eine grobe Skizze haben, eine Mitbewerberreferenz, oder einfach eine Marktlücke, die Sie schließen möchten – wir können daraus ein regalfertiges Produkt machen.
Welche Arten von LED-Schönheitsgeräten können durch OEM/ODM entwickelt werden??
Unsere OEM/ODM-Fähigkeiten decken eine breite Palette LED-basierter Schönheits- und Wellnessgeräte ab, einschließlich, aber nicht beschränkt auf:
- LED-Gesichtsmasken (Vollgesicht und Halbgesicht)
- LED-Therapiegürtel (auf Silikonbasis und OK-Tuchbasis)
- Rotlichttherapie-Ganzkörpermatten
- Multifunktionsgeräte Kombination von Lichttherapie und Vibration, Hitze, oder EMS
- Handgeführte LED-Behandlungsgeräte
- LED-Therapiepanels und gezielte Pads
Wir unterstützen mehrere Wellenlängenkonfigurationen, einschließlich 630 nm (Rot) und 850 nm (Nir), mit Optionen für zusätzliche Wellenlängen je nach beabsichtigter Anwendung. Wenn Sie ein Produktkonzept außerhalb dieser Kategorien haben, Kontaktieren Sie uns – unser R&Das D-Team bewertet regelmäßig neue Produkttypen.
Welche F&E- und Engineering-Fähigkeiten bietet Ihr Team??
Wakelife wird von Shenzhen Sungrow Led Technology Co. unterstützt., Ltd., mit fundiertem Fachwissen über den gesamten Produktentwicklungszyklus hinweg. Unsere internen Fähigkeiten umfassen:
- Industriedesign — Formfaktor, Ergonomie, Materialauswahl
- Optische Technik — LED-Array-Design, Wellenlängenauswahl, Optimierung der Bestrahlungsstärke
- Elektrotechnik — PCB-Design, Treiberschaltungen, Steuerungssysteme
- Prototyping & Validierung — EVT (Technischer Validierungstest), TVT (Designvalidierungstest), PVT (Produktionsvalidierungstest)
- Werkzeuge & Schimmelbildung — Eigene oder verwaltete Werkzeuge für kundenspezifische Gehäuse und Komponenten
- Medizinische Compliance — Bewertung des Regulierungspfads, Risikomanagement, und Zertifizierungsunterstützung vom ersten Tag an
Diese integrierte Teamstruktur bedeutet, dass Ihr Projekt unter einem Dach bleibt – wodurch Missverständnisse vermieden werden, Vorlaufzeit, und Kosten.
Was ist das MOQ für White Label?, OEM, und vollständige ODM-Projekte?
MOQ hängt von der Produktkategorie ab, Anpassungsebene, Schlüsselkomponenten, und Verpackungsanforderungen:
| Servicelevel | Typischer MOQ-Bereich | Warum |
|---|---|---|
| Weißes Etikett | Untere | Verwendet vorhandene Produktplattform und Tools |
| OEM | Medium | Möglicherweise sind Änderungen auf Komponentenebene erforderlich |
| Vollständiges ODM | Höher | Beinhaltet neue Werkzeuge, kundenspezifische Teile, und Validierungsaufwand |
Basierend auf Ihrem Zielmarkt und Ihrer Umsatzprognose schlagen wir Ihnen den am besten geeigneten MOQ-Plan vor. Für qualifizierte Projekte, wir können auch unterstützen Pilotläufe oder abgestufte Produktionspläne um Ihnen dabei zu helfen, den Markt zu testen, bevor Sie sich zu umfassenden Bestellungen verpflichten.
Fordern Sie ein Angebot an mit Ihren Projektdetails und wir geben Ihnen eine konkrete MOQ-Empfehlung.
Welche LED-Wellenlängen können in einem OEM/ODM-Projekt verwendet werden??
Unser R&Das D-Team unterstützt die vollständige Wellenlängenspezifikation in OEM/ODM-Projekten. Egal, ob Sie ein Produkt aus einer bestehenden Idee entwickeln oder von Grund auf neu bauen, aus denen Sie auswählen können 10 klinisch untersuchte Wellenlängen vom sichtbaren Blau bis zum tiefen Infrarot:
| Wellenlänge | Lichttyp | Hauptvorteile |
|---|---|---|
| 415 nm | Blau-Violett | Klinisch eingesetzt in der Phototherapie bei Gelbsucht (einschließlich Neugeborenengelbsucht); wirkt entzündungshemmend und antibakteriell auf der Haut |
| 460 nm | Blau | Blaues Licht mit schmalem Spektrum und mäßiger Gewebedurchdringung; hilft bei der Regulierung des Tagesrhythmus, Entzündungen reduzieren, tötet Akne verursachende Bakterien ab, ruhige Rötung, Ödeme lindern, und überschüssigen Talg kontrollieren |
| 590 nm | Gelb | Hilft, Pigmentierung und dunkle Flecken zu verblassen, Hautton aufhellen, glatte feine Linien, und unterstützen die Immunfunktion |
| 633 nm | Rot | Fördert die Aufhellung der Haut, reduziert Pigmentierung, verjüngt die Hautstruktur, glättet feine Fältchen, minimiert Poren, und stimuliert die Kollagenproduktion |
| 660 nm | Rot | Unterstützt die Schmerzlinderung, verbessert die Kollagenablagerung, reduziert belastungsbedingte Müdigkeit, lindert neuropathische Schmerzen und Entzündungen, und beschleunigt die Wundheilung |
| 810 nm | Nahinfrarot (Nir) | Fördert das Haarwachstum, verbessert die Genesung nach traumatischen Verletzungen, unterstützt die Schlaganfall-Rehabilitation, und beschleunigt die Wundheilung |
| 830 nm | Nahinfrarot (Nir) | Beschleunigt die Gewebeheilung, reduziert das Infektionsrisiko, stimuliert die Endorphinausschüttung, und unterstützt die Knochenreparatur |
| 850 nm | Nahinfrarot (Nir) | Entzündungshemmende Wirkung, fördert die Wundheilung der Haut, reduziert feine Linien, Falten, und Hyperpigmentierung, und fördert die Muskelregeneration |
| 1064 nm | Tiefes Infrarot | Bekämpft pigmentierte Hauterkrankungen, Entfernung von Tätowierungen und Permanent Make-up, Haarentfernung, Behandlung von Aknenarben, statische Reduzierung von Gesichtsfalten, und Fettauflösung |
| 1072 nm | Tiefes Infrarot | Erhöht die Filaggrin- und Aquaporin-Expression in Keratinozyten; steigert Prokollagen, Kollagen Typ I, und Elastinsynthese in Fibroblasten – unterstützt die Aufrechterhaltung der Hautbarriere, Anti-Aging, und Faltenreduktion |
Für vollständige OEM/ODM-Projekte, Die Wellenlängenauswahl ist nicht auf bestehende Produktkonfigurationen beschränkt – Sie können völlig neue Kombinationen definieren, die auf Ihre klinische Positionierung zugeschnitten sind, Zielmarkt, und Angaben zur beabsichtigten Verwendung.
Unsere Produkte nutzen fortschrittliche Dual-Core-Dual-Wave-LED-Chips Und Triple-Chip-Technologie, Ermöglicht mehrere Wellenlängen in einem einzigen Gerät. Jede LED kann bis zu liefern 3W pro Chip mit Bestrahlungsstärken erreichen 60+ MW/cm² an der Behandlungsoberfläche.
Unser medizinisches Compliance-Team ist vom ersten Tag an beteiligt, um zu beurteilen, welche Auswirkungen Ihre ausgewählte Wellenlängenkonfiguration hat Regulierungsweg, Angaben zur beabsichtigten Verwendung, Risikoklassifizierung, und Zertifizierungsanforderungen — Sicherstellen, dass das Produkt von Anfang an auf Konformität ausgelegt ist, Nacharbeit und spätere Kosten werden reduziert.
Alle Wellenlängenkonfigurationen durchlaufen EVT/DVT/PVT-Validierung für optische Leistung, Gleichmäßigkeit der Bestrahlungsstärke, Wärmemanagement, elektrische Sicherheit, und Biokompatibilität vor der Massenproduktion.
Was ist der typische Entwicklungszeitplan und was sind die wichtigsten Meilensteine??
Die Zeitpläne variieren je nach Projektkomplexität und Zertifizierungsumfang. Ein typisches OEM/ODM-Projekt folgt diesen Phasen:
| Phase | Beschreibung |
|---|---|
| 1. Konzept & Anforderungsausrichtung | Produktumfang definieren, Zielspezifikationen, Marktpositionierung |
| 2. Industriell & Technisches Design | ID/MD/EE-Design, optische Gestaltung, Stücklistenplanung |
| 3. Prototyp-Iterationen | Funktionsmuster zur Überprüfung und zum Testen |
| 4. EVT / TVT / PVT-Validierung | Maschinenbau, Design, und Produktionsvalidierung |
| 5. Werkzeuge | Formenentwicklung und Erstmusterprüfung |
| 6. Pilotlauf | Kleinserienfertigung zur endgültigen Qualitätsbestätigung |
| 7. Hochlauf zur Massenproduktion | Komplette Fertigung und Lieferung |
Nach der ersten Anforderungsprüfung, Wir erstellen einen detaillierten Projektplan mit Meilensteinen und Ergebnissen. Projekte, die eine behördliche Zertifizierung erfordern (FDA, Ce) beinhaltet zusätzliche Zeit für die Compliance-Dokumentation und -Tests.
Können Sie behördliche Zertifizierungen unterstützen? (FDA, Ce, TGA) für OEM/ODM-Produkte?
Ja. Unsere Muttergesellschaft verfügt über FDA 510K, Mdl, und TGA-Zertifizierungen, und wir haben ein engagiertes Medical-Compliance-Team, das sich engagiert von der Produktdesignphase an.
Ihre Rolle umfasst:
- Beurteilung des Regulierungspfads basierend auf Ihrem Zielmarkt
- Bestimmung des Verwendungszwecks und zulässiger Ansprüche
- Durchführung von Risikomanagementanalysen
- Wichtige Sicherheits- und Leistungsanforderungen frühzeitig erkennen
Das “Compliance-by-Design” Dieser Ansatz stellt sicher, dass das Produkt von Anfang an so gebaut ist, dass es die Zertifizierungsanforderungen erfüllt – und reduziert so Nacharbeiten, kosten, und Time-to-Market. Ob Sie eine FDA-Zulassung für den US-Markt benötigen, CE-Kennzeichnung für Europa, oder TGA-Registrierung für Australien, Wir können den Prozess begleiten und unterstützen.
Wie gehen Sie mit dem IP-Eigentum um?, Designdateien, und NDA-Schutz?
Wir befolgen ein strenges Vertraulichkeitsmanagement. Ein Eine NDA kann vor Beginn der Projektbesprechungen unterzeichnet werden.
Für ODM-Projekte, Folgendes legen wir in der Projektvereinbarung klar fest:
- IP-Eigentum — Wem gehört das Produktdesign und das damit verbundene geistige Eigentum?
- Designdateien — Eigentums- und Übertragungsbedingungen für CAD-Dateien, technische Zeichnungen, Stückliste
- Nutzungsrechte — Lizenzumfang, falls zutreffend
Ihre Marke und Ihre langfristigen Geschäftsinteressen bleiben während des gesamten Engagements geschützt. Wenn Sie spezielle IP-Bedenken haben, Bringen Sie sie frühzeitig in der Diskussion zur Sprache und wir werden die Vereinbarung entsprechend strukturieren.
Wer trägt die Werkzeug- und Formenkosten??
Bei kompletten ODM- und kundenspezifischen OEM-Projekten trägt der Kunde in der Regel die Werkzeug- und Formenkosten, da die Formen speziell für Ihr Produkt entwickelt werden. Jedoch, Die Vereinbarung richtet sich nach dem Projektumfang und der Auftragsbindung:
- Spezielle Formen — Der Kunde trägt die Werkzeugkosten; Formen können ausschließlich Eigentum des Auftraggebers sein
- Geteilte Plattformformen — Wenn Änderungen an einer bestehenden Produktplattform vorgenommen werden, Die Werkzeugkosten können deutlich niedriger sein oder teilweise übernommen werden
- Volumenversatz — In manchen Fällen, Werkzeugkosten können über Produktionsaufträge hinweg amortisiert werden
Wir werden eine transparente Aufschlüsselung der Werkzeugkosten in den Projektvorschlag aufnehmen, damit es keine Überraschungen gibt. Fordern Sie ein detailliertes Angebot für Ihr spezifisches Projekt an.
Können Sie eine APP-Steuerung oder intelligente Funktionen in das Gerät integrieren??
Ja. Wir können intelligente Funktionen entwickeln oder in Ihr OEM/ODM-Gerät integrieren, einschließlich:
- Bluetooth-APP-Steuerung — Auswahl des Behandlungsmodus, Timer, Intensitätsanpassung
- Voreingestellte Behandlungsprotokolle — Geführte Sitzungen je nach Hautproblem oder Körperregion
- Nutzungsverfolgung — Sitzungsverlauf, Erinnerungen, Compliance-Verfolgung
- Firmware-Updates – OTA (Over-the-Air) Updatefähigkeit
Egal, ob Sie eine Marken-Begleit-App oder die Integration in eine bestehende Plattform wünschen, Unser Elektronik-Engineering-Team kann die Hardware und Firmware zur Unterstützung entwerfen. Besprechen Sie Ihre Anforderungen an intelligente Funktionen frühzeitig in der Konzeptphase, um eine nahtlose Integration sicherzustellen.
Wie groß ist Ihre Fertigungskapazität, um eine Scale-up-Produktion zu unterstützen??
Wir betreiben zwei Produktionsstätten mit einer Gesamtfläche von 20,000 m² (Fabrik 1: 14,000 m²; Fabrik 2: 6,000 m²). Unsere Produktionsinfrastruktur unterstützt:
- Produkte erfordern FDA 510K, Mdl, und TGA Einhaltung
- Multifunktionsgeräte, die LED-Therapie mit Vibration kombinieren, Hitze, und andere Modalitäten
- Flexible Produktionsplanung – von der Nullserie bis zum Großauftrag
Die Kapazitätsplanung ist Teil des Projektvorschlags. Sobald Ihr Produkt die PVT-Validierung besteht, Wir erstellen einen Anlaufplan, der auf Ihren Startzeitplan und Ihre Bedarfsprognose abgestimmt ist.
Wie starte ich ein OEM/ODM-Projekt mit Wakelife??
Der Einstieg ist unkompliziert:
| Schritt | Was geschieht |
|---|---|
| Schritt 1 – Greifen Sie zu | Kontaktieren Sie uns mit Ihrer Produktidee, Referenz, oder kurz |
| Schritt 2 – NDA & Erstes Treffen | Bei Bedarf unterzeichnen wir eine NDA, Vereinbaren Sie dann ein Treffen, um Ihr Konzept und Ihre Ziele zu verstehen |
| Schritt 3 - Vorschlag & Zeitleiste | Wir liefern einen klaren Vorschlag mit Weitblick, Zeitleiste, MOQ, und Kostenvoranschlag |
| Schritt 4 - Design & Entwicklung | Vollständiger Entwicklungszyklus vom Konzept bis zum validierten Prototyp |
| Schritt 5 — Werkzeuge & Pilotlauf | Schimmelbildung, Pilotproduktion, endgültige Qualitätsbestätigung |
| Schritt 6 – Massenproduktion & Lieferung | Hochlauf zur Serienfertigung |
Für den Anfang benötigen Sie kein fertiges Design. Ganz gleich, ob Sie ein detailliertes Datenblatt haben oder einfach nur eine Marktchance im Auge haben, Unser Team begleitet Sie durch jede Phase.
Hey, Zögern Sie nicht, es zu versuchen & Fühlen Sie Muster, bevor Sie Ihre Entscheidung erfinden