ODM/OEM
- Monthly Production of 180k
- Customize the perfect LED beauty mask for your brand in as fast as 7 days.

Bring Your Vision to Life
Leverage our cutting-edge R&D and design expertise to transform your unique ideas into reality. Share your concept, and we’ll deliver a customized solution tailored to your needs.

Seamless Customization, Start to Finish
Our dedicated R&D, engineering, and design teams collaborate closely with you to craft the perfect LED beauty device. From initial concept to final product, we ensure a smooth, efficient process with rapid turnaround times.

Mass Production with Efficiency
With a team of 150+ skilled professionals and state-of-the-art automated production lines, we excel in handling large-scale orders. Our streamlined processes ensure timely delivery, helping you bring your products to market faster.

24/7 Reliable Support
Our customer support team is available around the clock to address any post-sales needs, ensuring a seamless and stress-free experience for you and your customers.
What can we customize?
Our masks help you build a deep connection with consumers and create a trustworthy beauty brand.

Appearance Customization
- Pattern
- Shape
- LOGO
- Instruction Manual
- Color
- Packaging
Internal Components
- LED Chip Selection
- LED Bead Quantity and Arrangement
- FPC Board
Controller Customization
- APP Customization
- Controller Handle
Controller Customization
- Irradiance: 10-50mW/c㎡
- Wavelength: 300nm-2000nm
Our OEM/ODM Product Range
LED Beauty Device OEM/ODM Production Process
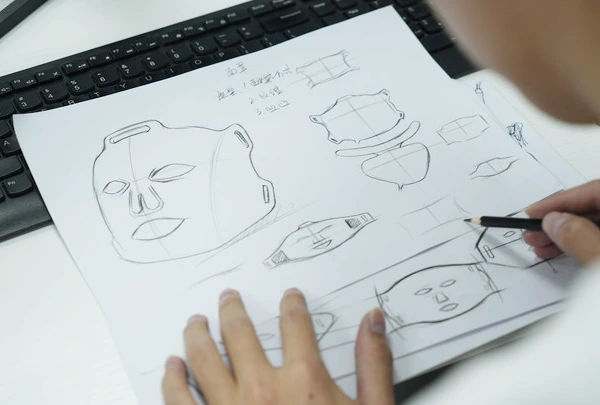
Appearance Design
Including patterns, shapes, logos, colors

Structural Design
Internal components such as LED bead chips, number of beads, FPC boards, etc.

3D Model Printing
Rapid generation of sample models through 3D printers

Label, Manual, and Packaging Design
Design your own branded labels, packaging and manuals.

Production Start
After completing the initial preparations, mass production begins.

Testing
The first batch of products is tested to ensure 100% compliance with quality standards.

Quality Inspection
Once mass production is completed, each product undergoes a thorough quality inspection.

Logistics & Shipping
Products are shipped via DHL, UPS, FedEx, TNT, EMS, and other reliable carriers.
Frequently Asked Questions About OEM/ODM LED Beauty Device Development
What is the difference between OEM and ODM, and how do they compare to other Wakelife services?
OEM (Original Equipment Manufacturing) means you provide the product design or specifications, and we manufacture it for you. ODM (Original Design Manufacturing) means we handle the full development — from concept and industrial design to engineering, prototyping, and mass production — based on your idea or brief.
Here’s how all four services compare:
- Wholesale — Purchase our existing products in bulk at competitive pricing. No customization involved.
- Private Label — Add your logo, packaging, and branding to our existing products. The product itself remains unchanged.
- Customization — Modify specific parameters (wavelength, power, treatment modes, colors) on an existing product platform.
- OEM/ODM — Develop a completely new or significantly modified product from the ground up, with full engineering, tooling, and compliance support.
If you’re unsure which path fits your business, contact our team and we’ll recommend the best approach based on your goals, budget, and timeline.
If I only have an idea, can Wakelife develop the product as a full ODM project?
Absolutely. We provide end-to-end ODM development — from product definition and industrial design to optical/electronic engineering, prototyping, validation, tooling, and mass production. You don’t need technical drawings or engineering files to get started.
Our team will align the design with your target market, brand positioning, and cost goals while ensuring manufacturability and compliance readiness. The process begins with a meeting to understand your concept, after which we deliver a clear proposal with a detailed timeline.
Whether you have a rough sketch, a competitor reference, or simply a market gap you want to fill — we can turn it into a shelf-ready product.
What types of LED beauty devices can be developed through OEM/ODM?
Our OEM/ODM capabilities cover a wide range of LED-based beauty and wellness devices, including but not limited to:
- LED face masks (full-face and half-face)
- LED therapy belts (silicone-based and OK Cloth-based)
- Red light therapy full body mats
- Multi-function devices combining light therapy with vibration, heat, or EMS
- Handheld LED treatment devices
- LED therapy panels and targeted pads
We support multiple wavelength configurations including 630nm (Red) and 850nm (NIR), with options for additional wavelengths depending on the intended application. If you have a product concept outside of these categories, reach out — our R&D team evaluates new product types regularly.
What R&D and engineering capabilities does your team offer?
Wakelife is backed by Shenzhen Sungrow Led Technology Co., Ltd., with deep expertise across the full product development cycle. Our in-house capabilities include:
- Industrial design — Form factor, ergonomics, materials selection
- Optical engineering — LED array design, wavelength selection, irradiance optimization
- Electronic engineering — PCB design, driver circuits, control systems
- Prototyping & validation — EVT (Engineering Validation Test), DVT (Design Validation Test), PVT (Production Validation Test)
- Tooling & mold development — In-house or managed tooling for custom housings and components
- Medical compliance — Regulatory pathway assessment, risk management, and certification support from day one
This integrated team structure means your project stays under one roof — reducing miscommunication, lead time, and cost.
What is the MOQ for white label, OEM, and full ODM projects?
MOQ depends on the product category, customization level, key components, and packaging requirements:
| Service Level | Typical MOQ Range | Why |
|---|---|---|
| White Label | Lower | Uses existing product platform and tooling |
| OEM | Medium | May require component-level changes |
| Full ODM | Higher | Involves new tooling, customized parts, and validation workload |
We will propose the most suitable MOQ plan based on your target market and sales forecast. For qualified projects, we can also support pilot runs or staged production plans to help you test the market before committing to full-scale orders.
Request a quote with your project details and we’ll provide a specific MOQ recommendation.
What LED wavelengths can be used in an OEM/ODM project?
Our R&D team supports full wavelength specification in OEM/ODM projects. Whether you are developing a product from an existing idea or building from scratch, you can select from 10 clinically studied wavelengths spanning visible blue to deep infrared:
| Wavelength | Light Type | Primary Benefits |
|---|---|---|
| 415 nm | Blue-Violet | Clinically used in jaundice phototherapy (including neonatal jaundice); provides anti-inflammatory and antibacterial effects on the skin |
| 460 nm | Blue | Narrow-spectrum blue light with moderate tissue penetration; helps regulate circadian rhythm, reduce inflammation, kill acne-causing bacteria, calm redness, relieve edema, and control excess sebum |
| 590 nm | Yellow | Helps fade pigmentation and dark spots, brighten skin tone, smooth fine lines, and support immune function |
| 633 nm | Red | Promotes skin brightening, reduces pigmentation, rejuvenates skin texture, smooths fine wrinkles, minimizes pores, and stimulates collagen production |
| 660 nm | Red | Supports pain relief, enhances collagen deposition, reduces exercise-induced fatigue, alleviates neuropathic pain and inflammation, and accelerates wound healing |
| 810 nm | Near-Infrared (NIR) | Promotes hair growth, improves recovery from traumatic injuries, supports stroke rehabilitation, and accelerates wound healing |
| 830 nm | Near-Infrared (NIR) | Accelerates tissue healing, reduces infection risk, stimulates endorphin release, and supports bone repair |
| 850 nm | Near-Infrared (NIR) | Anti-inflammatory benefits, promotes skin wound healing, reduces fine lines, wrinkles, and hyperpigmentation, and enhances muscle recovery |
| 1064 nm | Deep Infrared | Targets pigmented skin conditions, tattoo and permanent makeup removal, hair removal, acne scar treatment, static facial wrinkle reduction, and fat dissolution |
| 1072 nm | Deep Infrared | Increases filaggrin and aquaporin expression in keratinocytes; boosts procollagen, type I collagen, and elastin synthesis in fibroblasts — supports skin barrier maintenance, anti-aging, and wrinkle reduction |
For full OEM/ODM projects, wavelength selection is not limited to existing product configurations — you can define entirely new combinations tailored to your clinical positioning, target market, and intended use claims.
Our products utilize advanced dual-core dual-wave LED chips and triple chip technology, enabling multiple wavelengths within a single device. Each LED can deliver up to 3W per chip with irradiance levels reaching 60+ mW/cm² at the treatment surface.
Our medical compliance team is involved from day one to assess how your selected wavelength configuration impacts regulatory pathway, intended use claims, risk classification, and certification requirements — ensuring the product is designed for compliance from the beginning, reducing rework and cost later.
All wavelength configurations undergo EVT/DVT/PVT validation for optical performance, irradiance uniformity, thermal management, electrical safety, and biocompatibility before mass production.
What is the typical development timeline and what are the key milestones?
Timelines vary by project complexity and certification scope. A typical OEM/ODM project follows these stages:
| Phase | Description |
|---|---|
| 1. Concept & Requirements Alignment | Define product scope, target specs, market positioning |
| 2. Industrial & Engineering Design | ID/MD/EE design, optical layout, BOM planning |
| 3. Prototype Iterations | Functional samples for review and testing |
| 4. EVT / DVT / PVT Validation | Engineering, design, and production validation |
| 5. Tooling | Mold development and first article inspection |
| 6. Pilot Run | Small-batch production for final quality confirmation |
| 7. Ramp-Up to Mass Production | Full-scale manufacturing and delivery |
After the initial requirement review, we provide a detailed project plan with milestones and deliverables. Projects requiring regulatory certification (FDA, CE) will include additional time for compliance documentation and testing.
Can you support regulatory certifications (FDA, CE, TGA) for OEM/ODM products?
Yes. Our parent company holds FDA 510K, MDL, and TGA certifications, and we have a dedicated medical compliance team that gets involved from the product design stage.
Their role includes:
- Assessing the regulatory pathway based on your target market
- Defining intended use and permissible claims
- Conducting risk management analysis
- Identifying key safety and performance requirements early
This “compliance-by-design” approach ensures the product is built to meet certification requirements from the beginning — reducing rework, cost, and time-to-market. Whether you need FDA clearance for the US market, CE marking for Europe, or TGA registration for Australia, we can guide and support the process.
How do you handle IP ownership, design files, and NDA protection?
We follow strict confidentiality management. An NDA can be signed before any project discussions begin.
For ODM projects, we clearly define the following in the project agreement:
- IP ownership — Who owns the product design and related intellectual property
- Design files — Ownership and transfer terms for CAD files, engineering drawings, BOM
- Usage rights — Licensing scope if applicable
Your brand and long-term business interests are protected throughout the engagement. If you have specific IP concerns, raise them early in the discussion and we’ll structure the agreement accordingly.
Who pays for tooling and mold costs?
Tooling and mold costs are typically the client’s responsibility for full ODM and custom OEM projects, as the molds are developed specifically for your product. However, the arrangement depends on the project scope and order commitment:
- Dedicated molds — Client pays tooling cost; molds can be exclusively owned by the client
- Shared platform molds — If modifications are made to an existing product platform, tooling costs may be significantly lower or partially absorbed
- Volume offset — In some cases, tooling costs can be amortized across production orders
We will include a transparent tooling cost breakdown in the project proposal so there are no surprises. Request a detailed quote for your specific project.
Can you integrate APP control or smart features into the device?
Yes. We can develop or integrate smart features into your OEM/ODM device, including:
- Bluetooth APP control — Treatment mode selection, timer, intensity adjustment
- Pre-set treatment protocols — Guided sessions based on skin concern or body area
- Usage tracking — Session history, reminders, compliance tracking
- Firmware updates — OTA (over-the-air) update capability
Whether you want a branded companion app or integration with an existing platform, our electronic engineering team can design the hardware and firmware to support it. Discuss your smart feature requirements early in the concept phase to ensure seamless integration.
What is your manufacturing capacity to support scale-up production?
We operate two manufacturing facilities with a combined area of 20,000 m² (Factory 1: 14,000 m²; Factory 2: 6,000 m²). Our production infrastructure supports:
- Products requiring FDA 510K, MDL, and TGA compliance
- Multi-function devices combining LED therapy with vibration, heat, and other modalities
- Flexible production planning — from pilot runs to large-scale orders
Capacity planning is part of the project proposal. Once your product passes PVT validation, we establish a ramp-up schedule aligned with your launch timeline and demand forecast.
How do I start an OEM/ODM project with Wakelife?
Getting started is straightforward:
| Step | What Happens |
|---|---|
| Step 1 — Reach Out | Contact us with your product idea, reference, or brief |
| Step 2 — NDA & Initial Meeting | We sign an NDA if needed, then schedule a meeting to understand your concept and goals |
| Step 3 — Proposal & Timeline | We deliver a clear proposal with scope, timeline, MOQ, and cost estimate |
| Step 4 — Design & Development | Full development cycle from concept to validated prototype |
| Step 5 — Tooling & Pilot Run | Mold development, pilot production, final quality confirmation |
| Step 6 — Mass Production & Delivery | Ramp-up to full-scale manufacturing |
You don’t need a finished design to start. Whether you have a detailed spec sheet or just a market opportunity in mind, our team will guide you through every stage.
Hey, don’t hesitate to try & feel SAMPLES before making up your decision